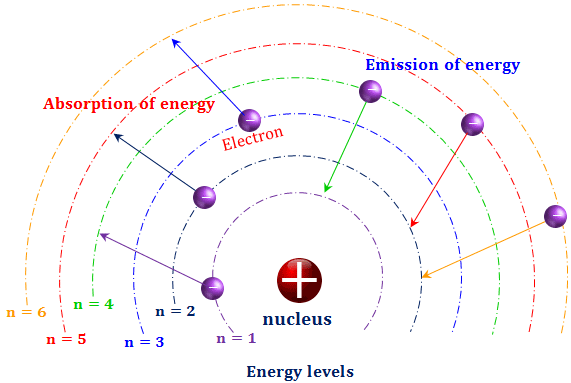
That is a reason not to teach about circular orbits and L = nħ. But second, it doesn’t conform to the essence of what Bohr actually did. This, in itself, is not a reason to scrap it, for the historical development of quantum physics is certainly of interest. In 1913, the physicist Niels Bohr introduced a model of the atom that contributed a greater understanding to its structure and quantum mechanics. First it doesn’t conform to our present understanding of the hydrogen atom. This picture of the hydrogen atom is wrong in two senses. Its title was Het Atommodel van Rutherford-Bohr. The vast majority of its mass is located in a positively charged central nucleus, which is 10,000. Rutherford had made the startling discovery that most of the atom is empty space. The Bohr model works for one-electron atoms or ions only because certain factors present in more complex atoms are not present in these atoms or ions. Burgers, a student of Ehrenfest’s in Leiden, wrote his doctoral dissertation on Bohr’s new atomic theory, probably the first dissertation of its kind. Bohr, one of the pioneers of quantum theory, had taken the atomic model presented a few years earlier by physicist Ernest Rutherford and given it a quantum twist. 
This theory built on the earlier work of Rutherford and others and provided a more detailed understanding of the behavior of electrons within the atom. Electrons in smaller orbits will have less energy as a result. To summarise Bohr’s atomic model, electron energy levels are focussed on the size of the orbits. This “pedagogic” version postulates that an electron in a stationary state moves in a circular orbit with an angular momentum that is an integral multiple of h/2π ( L = nh/2π = nħ)- ħ for the lowest-energy state, 2 ħ for the next state, and so on. The progress and consolidation of the Bohr atom in the decade after its birth may be illustrated in various ways. Bohr’s Atomic Theory, also known as the Bohr Model of the Atom, was developed by Niels Bohr in 1913, following his work on the structure of the hydrogen atom. In Niels Bohr’s atomic theory, electrons with fixed sizes and energies travel in orbits around a positively charged nucleus, similar to how planets orbit the sun. Unfortunately, the version of the Bohr atom that appears in many textbooks and is no doubt often presented to students is more wrong than right and may leave both teachers and students wondering why, more than a hundred years later, it is still being taught. This quiz delves into atomic structures, electron configurations, and the Bohr models depiction of the atom as a miniature solar system. 
This is an excellent idea, given the historic importance of Bohr’s 1913 work, which provided the bridge from Planck’s quantized interaction of matter and radiation (1900) to the full theory of quantum mechanics (1925-28). Have you explored Bohr-Rutherford diagrams in your Chemistry studies Test your knowledge with our Bohr Diagram Quiz Questions and Answers. Many teachers like to introduce the Bohr atom toward the end of an introductory physics course. This video looks at the pioneering work of Niels Bohr who proposed a novel model of the atom in 1913 which would lay the foundations for a quantum mechanical.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
